 Sections were deparaffinized in xylene and rehydrated in a graded alcohol series. Taken together, we demonstrate that pathological hypertrophy occurs in AC and is secondary to cardiomyocyte loss and cardiac remodeling. Calcineurin/nuclear factor of activated T cells (Nfat) signaling, which is linked to pathological hypertrophy, is observed during AC progression, as evidenced by Nfatc2 and Nfatc3 mRNA in cardiomyocytes and increased mRNA of the Nfat target regulator of calcineurin 1. In parallel, mRNA expression of myosin heavy chain 7 and natriuretic peptide B is up-regulated in both ventricles with right ventricular preference. Hypertrophy progresses further during the chronic disease stage. In desmoglein 2-mutant mice, cardiomyocyte diameters are normal during the concealed disease phase but increase significantly after acute disease onset on cardiomyocyte death and fibrotic myocardial remodeling. Elevated myosin heavy chain 7 mRNA expression is detected in left ventricles. Increased mRNA expression of the cardiac stress marker natriuretic peptide B is also observed in the right ventricle of AC patients. We find that cardiomyocyte diameters are significantly increased in right ventricles of AC patients. To this end, we examined tissue samples from AC patients with end-stage heart failure and tissue samples that were collected at different disease stages from desmoglein 2-mutant mice, a well characterized AC model. Our goal was to determine whether cardiomyocytes respond to AC progression by pathological hypertrophy. 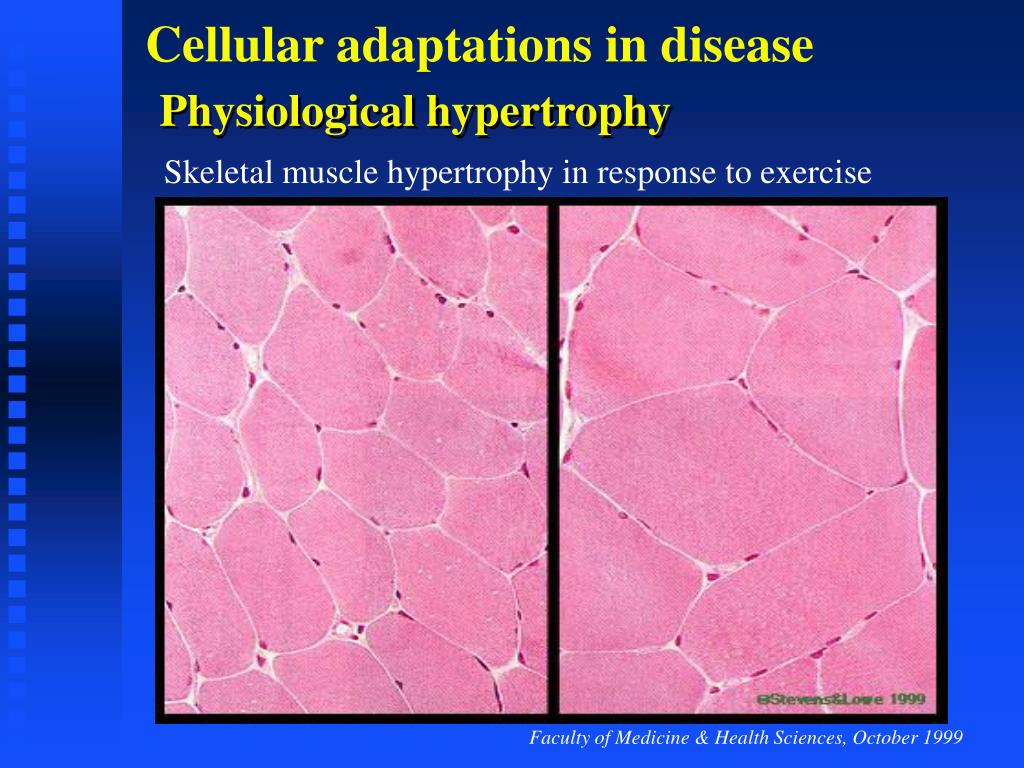
AC pathology is characterized by cardiomyocyte loss and replacement fibrosis. 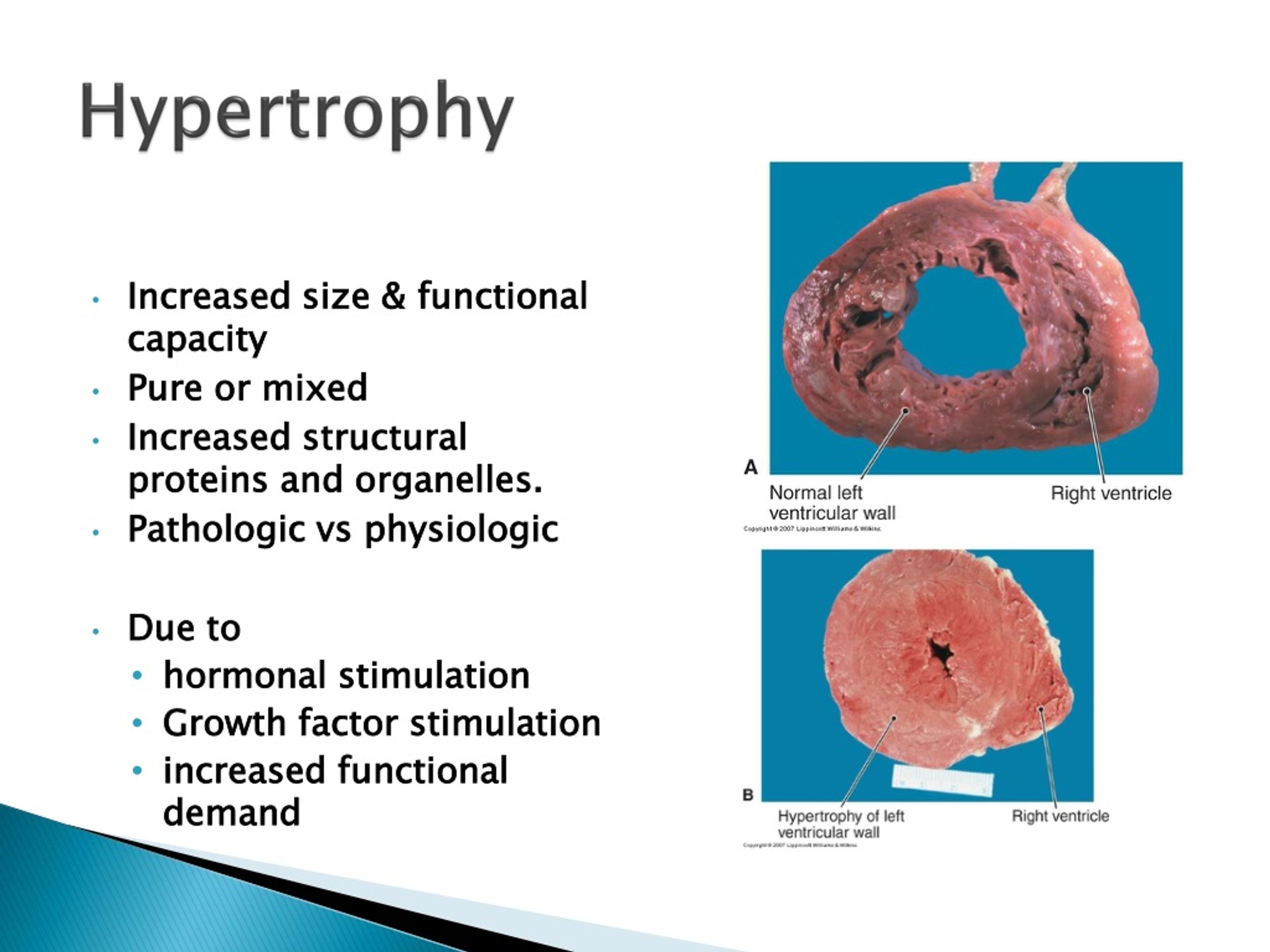
Arrhythmogenic cardiomyopathy (AC) is a hereditary disease leading to sudden cardiac death or heart failure. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
